
Boyoubei® is a biosimilar to Prolia®.
(This product is a prescription drug, please take this drug with doctor’s prescription and under doctor’s instruction)
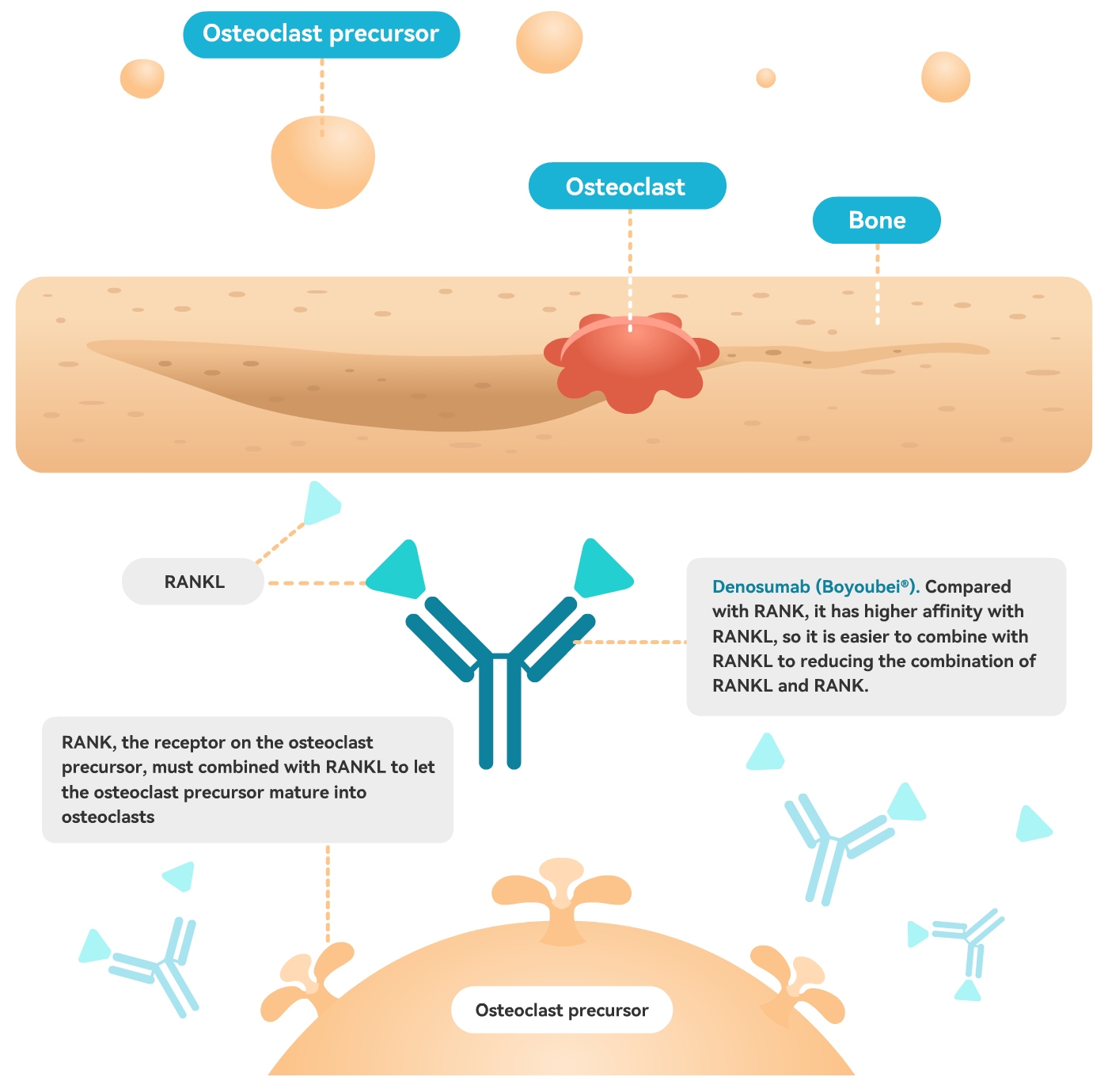
Osteoclasts are the cells that can degrade bone. In the case of hyperfunction, it will cause bone/joint diseases, including osteoporosis. The differentiation, maturation and activation of osteoclasts require RANKL (osteoclast differentiation factor) to bind to the receptor RANK on the surface of osteoclast precursors. Deshumumab combines with RANKL with high affinity, competitively reducing the combination of RANKL and RANK, making osteoclast precursors unable to differentiate and mature, reducing the generation of osteoclasts, thereby reducing bone absorption, increasing bone mass, improving bone strength, and achieving the goal of treating osteoporosis.
References:
1. Brown JP, et al. J Bone Miner Res 2009;24:153‒61.
2. Kendler DL, et al. J Bone Miner Res 2010;25:72‒81.
3. Roux C, et al. Bone 2014;58:48‒54.
4. Recknor C, et al. Obstet Gynecol 2013;121:1291‒9.
5. Miller PD, et al. J Clin Endocrinol Metab 2016;101:3163‒70.
